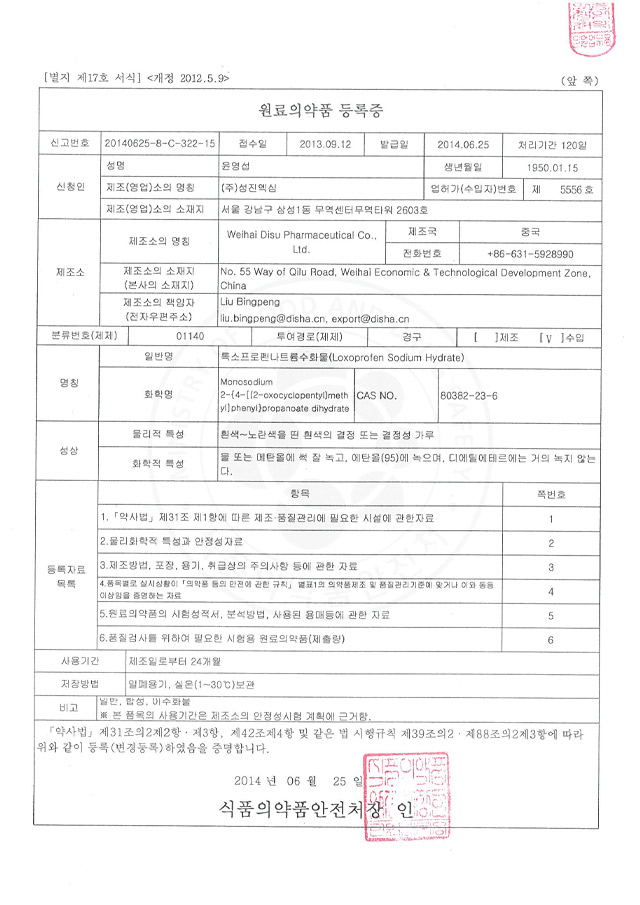
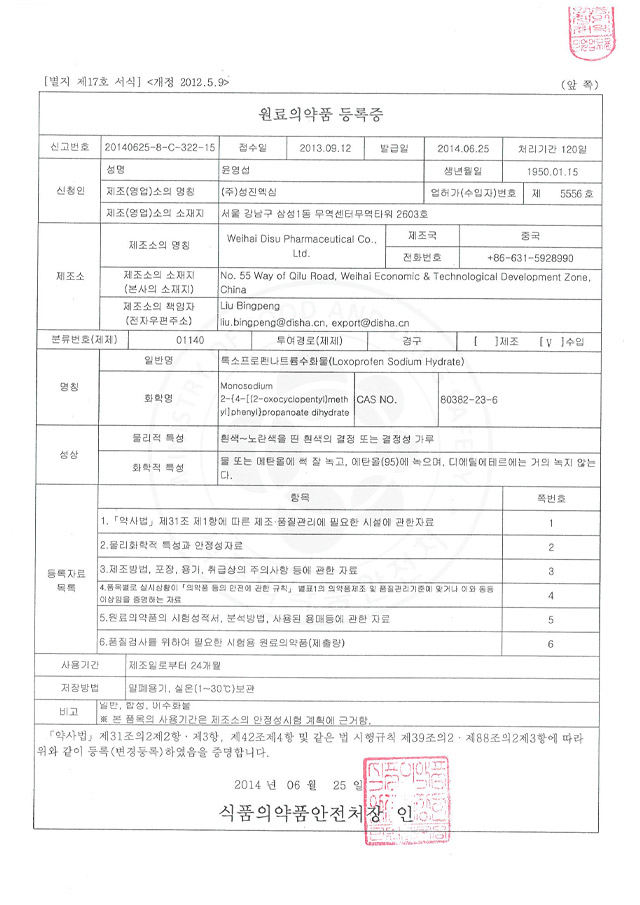
The company has a complete organizational structure and complete staffing. The management and technical personnel have corresponding professional knowledge and work experience, and can perform their respective duties in various management; they can organize production and carry out product quality control in accordance with GMP requirements.
Quality management and inspection personnel are subject to corresponding professional and technical training; the quality inspection center is equipped with places, instruments and equipment suitable for the scale, variety and inspection requirements of drug production; the quality management department is responsible for formulating internal control standards for materials, intermediate products and finished products, reviewing the handling procedures for unqualified products, and determining the release of finished products.